IBDP CORE>TOPIC 2.2> WATER
| Reaction_types.swf | |
| File Size: | 232 kb |
| File Type: | swf |
| Types of bond.swf | |
| File Size: | 1025 kb |
| File Type: | swf |
| watermolecule.swf | |
| File Size: | 1647 kb |
| File Type: | swf |
|
|
|
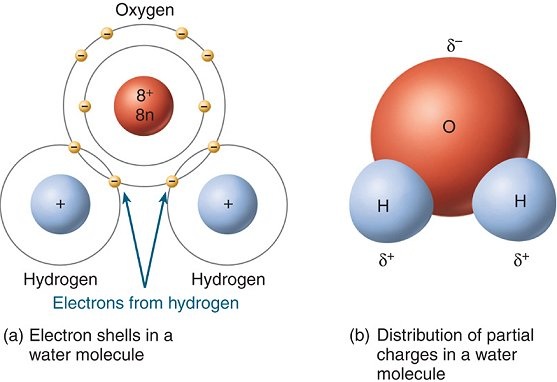
STRUCTURE OF WATER MOLECULE
Outline the thermal, cohesive and solvent properties of water.
Thermal properties-
Thermal capacity of water is high; therefore fluids containing water can carry away much heat;
The latent heat of vaporisation of water is high; water released onto the surface (sweat / saliva) carries away much heat by evaporation;
Cohesive properties-
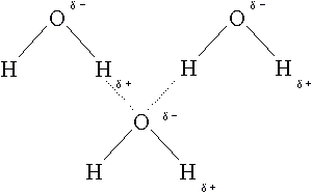
Water molecules are strongly cohesive (they tend to stick to one another)
Water molecules will also tend to stick to other molecules that are charged or polar (adhesion)
Hydrogen bonds between water molecules make them cohesive/stick together; this gives water a high surface tension / explains how water rises up xylem
Solvent properties-
Water molecules are polar; this makes water a good solvent.
Water can dissolve many organic and inorganic substances that contain electronegative atoms.
This occurs because the polar attraction of large quantities of water molecules can sufficiently weaken intramolecular forces (such as ionic bonds) and result in the dissociation of the atoms.
Thermal capacity of water is high; therefore fluids containing water can carry away much heat;
The latent heat of vaporisation of water is high; water released onto the surface (sweat / saliva) carries away much heat by evaporation;
Cohesive properties-
Water molecules are strongly cohesive (they tend to stick to one another)
Water molecules will also tend to stick to other molecules that are charged or polar (adhesion)
Hydrogen bonds between water molecules make them cohesive/stick together; this gives water a high surface tension / explains how water rises up xylem
Solvent properties-
Water molecules are polar; this makes water a good solvent.
Water can dissolve many organic and inorganic substances that contain electronegative atoms.
This occurs because the polar attraction of large quantities of water molecules can sufficiently weaken intramolecular forces (such as ionic bonds) and result in the dissociation of the atoms.
Explain the relationship between the properties of water and its uses in living organisms as a coolant, medium for metabolic reactions and transport medium.
Acts as a coolant
Thermal capacity of water is high; therefore fluids containing water can carry away much heat;
The latent heat of vaporisation of water is high; water released onto the surface (sweating and panting in animals and transpiration in plants) carries away much heat by evaporation;
As a transport medium;water dissolves many substances; (this facilitates metabolic reactions)
Water remains liquid over much of the range of the Earth’s temperatures; as a fluid, water is used as a medium to transport biological materials; e.g. blood / lymph / other suitable example;
Water can transport heat; water currents disperse larval forms of marine species / sperm / eggs; water as a habitat;
Water is densest at 4°C / water freezes at the surface first; water bodies are thermally stable;
Water is transparent / light sensitive receptors can operate under water;
Water has a high surface tension / some animals can walk / live on the surface film;
Thermal capacity of water is high; therefore fluids containing water can carry away much heat;
The latent heat of vaporisation of water is high; water released onto the surface (sweating and panting in animals and transpiration in plants) carries away much heat by evaporation;
As a transport medium;water dissolves many substances; (this facilitates metabolic reactions)
Water remains liquid over much of the range of the Earth’s temperatures; as a fluid, water is used as a medium to transport biological materials; e.g. blood / lymph / other suitable example;
Water can transport heat; water currents disperse larval forms of marine species / sperm / eggs; water as a habitat;
Water is densest at 4°C / water freezes at the surface first; water bodies are thermally stable;
Water is transparent / light sensitive receptors can operate under water;
Water has a high surface tension / some animals can walk / live on the surface film;
AQUA MINUTES AND KAHOOT!!!!!